Jingyu Zhang, Qian Deng, Maierhaba Maitiyaer, Li Wang, Amy Michelle Huang, Yue Liang, Qiudan Huang, ShuiLian Yu & Zhiping Liu
Abstract
Objective
To evaluate the relation between corneal optical quality and ocular surface manifestations in Chinese female patients with Sjogren’s syndrome dry eye (SSDE).
Methods
Cross-sectional study of female SSDE patients. Demographic information and ophthalmological and rheumatological indicators were collected. Ocular Surface Disease Index (OSDI) and Chinese Dry Eye Questionnaire (CDEQ), best-corrected visual acuity (BCVA), first and mean noninvasive tear break-up time (FNIBUT and MNIBUT), Schirmer I testing, Oxford Staining Score (OSS), meibomian gland loss (MGL), and optical quality were evaluated using generalized estimating equation (GEE) models. Multinomial logistic regression models and multiple linear regression models were employed to assess the correlations between dry eye indicators and corneal optical quality.
Results
27 SSDE patients (47 eyes), 9 Non-Sjögren’s Syndrome dry eye patients (NSSDE, 14 eyes), and 23 normal controls (NC, 44 eyes) were included. More severe dry eye signs and poorer results of corneal optical qualities were found in Chinese female SSDE patients (all p < 0.05). More severe dry eye signs (CDEQ score, FNIBUT, MNIBUT, OSS, Schirmer I test, lipid layer distribution, and MGL) and poorer results of corneal optical qualities (angle α) were found in SSDE patients (all p < 0.05). In addition, there was a significant difference in astigmatism (posterior corneal surface astigmatism and the types of astigmatism on the anterior and posterior surface of the cornea) between the groups (all p < 0.05).
Conclusions
Chinese females with DE, and particularly those with concurrent SS, demonstrated poorer ocular surface and corneal optical quality measures than those without DE.
Introduction
Dry eye (DE) disease is a common, chronic, inflammatory condition. According to previous studies, women are more susceptible to DE compared to men, and the prevalence increases with age 1. Dry eye (DE) is a common, chronic, inflammatory condition that affects the ocular surface and tear film, leading to discomfort, visual impairment, and reduced quality of life 2. This disease can trigger changes in the ocular surface and tear film 3,4, which in turn can increase ocular discomfort, fatigue, and visual impairment that can interfere with a patient’s daily activities, such as reading, driving, and electronics use, to varying degrees 1. DE disease can cause corneal damage, such as by inducing inflammation, protein and lipid deposition, corneal edema, and corneal neovascularization. This adversely affects corneal refraction and the optical pathway to the retina, and in severe cases, it can even lead to blindness 5. Currently, abnormal visual function is included in the definition and diagnostic criteria of DE disease by the Asia Dry Eye Society 6.
Certain systemic diseases, such as Sjogren’s syndrome (SS), are considered to be a risk factor for DE disease 7. This systemic autoimmune disease involves the exocrine glands and is primarily associated with immune cell dysregulation 8,9. The disease has an insidious onset and diverse clinical manifestations, with up to 98% of patients presenting with dryness of various organ systems 10. Many patients present with dry mouth and eyes due to decreased function of the salivary and lacrimal glands 11. SS, an autoimmune disease, is a significant risk factor for DE, often presenting with severe ocular surface manifestations due to decreased tear production and increased tear film instability 11. Tear film instability and ocular surface inflammation can lead to a vicious cycle that complicates the treatment of DE disease 12,13. If this disease is not treated in a timely manner, the patient’s symptoms may worsen, become more difficult to treat, and may lead to permanent ocular damage.
Previous studies have explored the impact of DE on corneal optical quality 14, but the relationship remains incompletely understood, particularly in Chinese female patients with SSDE. This study aims to fill this gap by evaluating the correlation between corneal optical quality and clinical ocular surface manifestations in Chinese female patients with SSDE. This research aims to elucidate the relationship between corneal optical quality and clinical ocular surface manifestations in Chinese females with Sjogren’s syndrome dry eye (SSDE), and to identify potential indicators for early intervention to preserve visual quality.
Methods
Study design and participants
This cross-sectional case–control study was performed according to the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Second Hospital of Guangzhou Medical University (2019-hs-12).
We recruited 27 SSDE patients (47 eyes), 9 Non-Sjögren’s Syndrome dry eye patients (NSSDE, 14 eyes), and 23 normal controls (NC, 44 eyes) from July 2021 to December 2023. The flowchart of participant screening is shown in Fig. 1. The diagnosis and classification of SS patients were identified by a rheumatologist (YSL) in accordance with the 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjogren’s syndrome 15. The inclusion criteria included the following: (1) aged 20–60 years; (2) intraocular pressure (IOP) < 21 mmHg; (3) no other systemic or ocular diseases. Exclusion criteria included the following: ocular anatomical abnormalities (eyelid entropion, eyelid scarring, etc.); history of glaucoma; contact lens wearers; intraocular surgery within the last 12 months; recent eye infection; and pregnant or lactating women. The sample size for the Non-Sjögren’s Syndrome dry eye (NSSDE) group was limited due to the specific inclusion criteria and the relative rarity of this subgroup among the overall dry eye population. While NSSDE patients are encountered in clinical practice, recruiting a larger sample size within the study timeframe was challenging. Future studies may focus on expanding this cohort to provide more robust data.
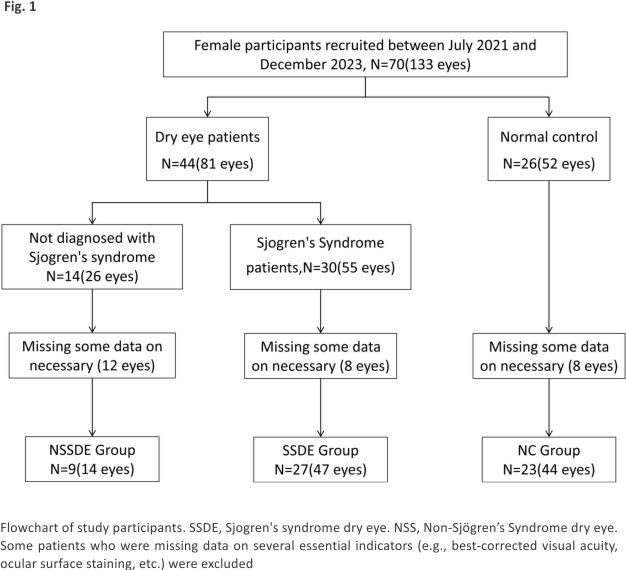 Patients were included based on the presence of clinically significant dry eye symptoms and signs in at least one eye. While dry eye disease (DED) often affects both eyes, particularly in patients with Sjogren’s syndrome, the severity and manifestations can vary between eyes. Therefore, both eyes were included if they met the inclusion criteria, while only one eye was included if the other did not meet the criteria or if the patient had unilateral disease.
Patients were included based on the presence of clinically significant dry eye symptoms and signs in at least one eye. While dry eye disease (DED) often affects both eyes, particularly in patients with Sjogren’s syndrome, the severity and manifestations can vary between eyes. Therefore, both eyes were included if they met the inclusion criteria, while only one eye was included if the other did not meet the criteria or if the patient had unilateral disease.
All the participants received a standardized ophthalmological examination consisting of best corrected visual acuity (BCVA), IOP, and slit-lamp examination at the time of enrollment.
Dry eye questionnaire collection
Participants were instructed to fill out the Ocular Surface Disease Index (OSDI) and the Chinese Dry Eye Questionnaire, which required them to describe the impact of their current symptoms on their daily lives. The OSDI ranged from 0–100, while the Chinese Dry Eye Questionnaire ranged from 0–48. An OSDI score of less than 20 is considered normal or mild dry eye; 20–45 is considered moderate; and more than 45 is considered severe dry eye 16. A CDEQ score of more than 7 is considered as symptomatic DE 17.
Ocular surface measures
We examined tear meniscus height (TMH), staining of the ocular surface, first and mean noninvasive tear break-up time (FNIBUT and MNIBUT), lipid layer condition, bulbar conjunctival hyperemia (BCH), and the condition of meibomian gland loss (MGL). The above subjects were measured and evaluated using a Keratograph 5 M (Oculus, Wetzlar, Germany), which has a high level of discrimination and diagnostic accuracy 18. The staining map obtained by Oculus was scored in conjunction with the Oxford Staining Score(OSS) to assess the damage to the ocular surface. In addition, the best corrected visual acuity (BCVA) and intraocular pressure (IOP) of all patients were tested before the ocular surface examination. Best-corrected visual acuity (BCVA) was measured and converted to LogMAR for analysis. Original measurements were taken using a standard ETDR chart. The conversion formula is:logMAR = log10 (1/visual acuity value)”.
Examinations were conducted in a controlled environment with a temperature of 22-25 ℃ and humidity of 40–60%. Participants were acclimated to the room for at least 15 min before testing. Ocular surface tests were performed in the following order: tear meniscus height (TMH), first and mean noninvasive tear break-up time (FNIBUT and MNIBUT), meibomian gland loss (MGL), ocular surface staining, and Schirmer I test. This sequence minimizes the impact of eyelid manipulation and fluorescein instillation on subsequent measurements. Lipid layer distribution was assessed using the Keratograph 5 M (Oculus, Wetzlar, Germany) and graded based on the presence and uniformity of the lipid layer. Meibomian gland loss was evaluated using the Meiboscore system validated by Reiko Arita 20.
Based on a previous study, fluorescein staining with the yellow filter has the advantage of simultaneous observation of both corneal and conjunctival damage in patients with dry eye without the need for additional vital staining 21. Corneal staining was assessed 2–3 min after fluorescein instillation to ensure stable conditions. The Schirmer I test was performed with the eyes closed to standardize tear production measurement. Fluorescein strips (Oculus, Wetzlar, Germany) and Schirmer test strips (Tianjin Jingming New Technology Development Co., Ltd., Tianjin, China) were used. The Schirmer I test without anaesthesia was used to measure tear production. A dry Schirmer test strip was inserted over the outer one-third of the lower eyelid margin, and the distance that the tears travelled along the test strip at 5 min was recorded as the Schirmer I score.
Corneal optical quality measures
The Pentacam (Oculus, Wetzlar, Germany) was used to assess corneal morphology, including astigmatism on the anterior and posterior surfaces. The iTrace wavefront aberrometer (Tracey Technologies, Houston, TX, USA) was used to directly measure corneal optical quality, including total corneal high-order aberrations (tHOAs), spherical aberrations (SAs), comas, modulation transfer function (MTF), and angles α and κ. These measurements provide a comprehensive evaluation of corneal optical quality.
Statistical analysis
The statistical power was calculated using PASS 15.0 software. Stata statistical software (V.17.0, Stata Corp, College Station, TX) was used for statistical analyses in this study, and p < 0.05 was considered a statistically significant difference. Means, standard errors, and weighted percentages were used for continuous variables, and frequency and weighted percentages were used for categorical variables. Generalized estimating equation (GEE) models were used to account for the intercorrelation of eyes within study subjects. Eyes (left or right) were set as within-subject variables in the GEE models. The measurements were dependent variables, while age and eye were set as covariates. Normality of distribution was verified using the Kolmogorov–Smirnov test. Given the small sample size of the NSSDE group, normality tests were performed on each dataset. For variables that did not follow a normal distribution, data were presented as median (interquartile range, Q1-Q3) to accurately reflect the central tendency and dispersion (Supplementary Table 1). Group comparisons for normally distributed continuous variables were performed using GEE. Kruskal–Wallis tests were utilized to analyze non-normally distributed data. Categorical data were compared using χ2 tests. Receiver operating characteristic (ROC) and area under the curve (AUC) were used to assess the predictive ability of statistically significant ocular surface indicators for DE disease. To explore the relationship between ocular surface indicators and corneal optical quality indicators and between-group differences, before conducting regression analysis, associations between dependent and independent variables were assessed using Kendall’s tau-b correlation coefficient. For multiple linear regression analysis, the dependent variable was continuous and normally distributed (e.g., corneal optical quality indicators). For multinomial logistic regression analysis, the dependent variable was categorical, following a binomial distribution (e.g., presence or absence of specific ocular surface manifestations).
Results
Clinical ocular surface manifestations and optical quality performance
Sample size calculation was performed based on the expected effect size. In our current study, a sample size of 30 eyes in the SSDE group, 10 eyes in the NSSDE group, and 30 eyes in the NC group achieved a statistical power of 93% in differentiating the Schirmer I test, OSS, FNIBUT, and MNIBUT measurements.
In this study, participants were recruited between July 2021 and December 2023 at the Second Affiliated Hospital of Guangzhou Medical University. 27 SSDE patients (47 eyes), 9 NSSDE patients (14 eyes), and 23 NC participants (44 eyes) met the inclusion and exclusion criteria. We performed further SS disease diagnosis and corneal optical quality assessment on these participants. After diagnosis by the same rheumatologist according to the 2016 American College of Rheumatology/ European League Against Rheumatism classification criteria for primary Sjogren’s syndrome, the above participants were enrolled in this study. Rheumatologic indicators of SSDE patients are shown in Table 1.
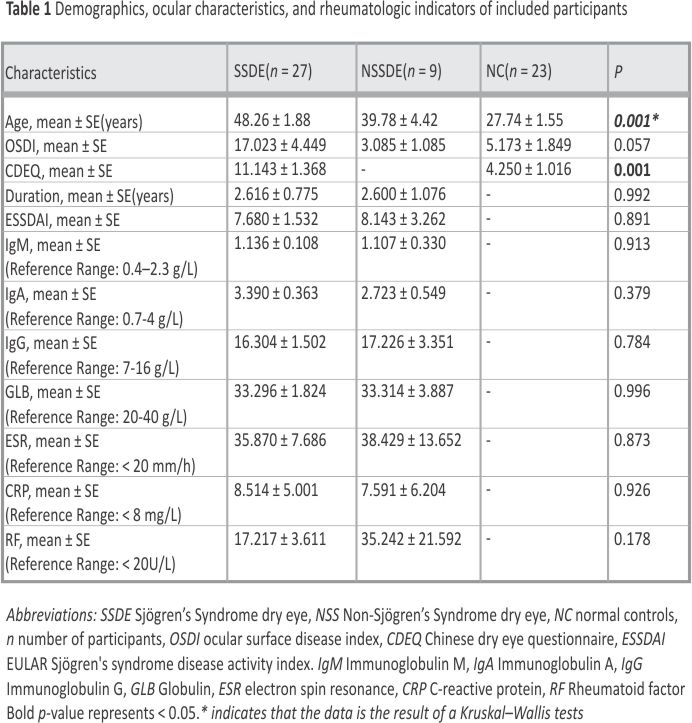 As shown in Table 2, compared to the NSSDE group and NC group, those with SSDE had decreased tear film breakup times (5.580 ± 0.529 s, p < 0.001; 9.186 ± 0.844 s, p = 0.001), and lower tear secretion (4.617 ± 0.579 mm/5 min, p < 0.001) over the same period of time. Uneven distribution of the lipid layer (p = 0.003) was observed in SSDE patients. Additionally, the upper MGL (p < 0.001) and lower MGL (p = 0.001) were worse in SSDE patients. The results of the participant questionnaire showed that the CDEQ scores of SSDE patients were higher than those of the NC group (11.143 ± 1.368, p = 0.001). BCVA was converted to the logarithmic minimum angle of resolution (logMAR). There was a statistically significant difference in logMAR between the three groups of participants (p = 0.001), which may have been due to the greater mean age of the SS group (p < 0.001). The ability of statistically different ocular surface indicators to discriminate SS can be reflected in Fig. 2. OSS, Schirmer I test, CDEQ, FNIBUT and MNIBUT all exhibit robust discriminatory capacity with AUCs of 0.962, 0.953, 0.861, 0,799 and 0.769, respectively.
As shown in Table 2, compared to the NSSDE group and NC group, those with SSDE had decreased tear film breakup times (5.580 ± 0.529 s, p < 0.001; 9.186 ± 0.844 s, p = 0.001), and lower tear secretion (4.617 ± 0.579 mm/5 min, p < 0.001) over the same period of time. Uneven distribution of the lipid layer (p = 0.003) was observed in SSDE patients. Additionally, the upper MGL (p < 0.001) and lower MGL (p = 0.001) were worse in SSDE patients. The results of the participant questionnaire showed that the CDEQ scores of SSDE patients were higher than those of the NC group (11.143 ± 1.368, p = 0.001). BCVA was converted to the logarithmic minimum angle of resolution (logMAR). There was a statistically significant difference in logMAR between the three groups of participants (p = 0.001), which may have been due to the greater mean age of the SS group (p < 0.001). The ability of statistically different ocular surface indicators to discriminate SS can be reflected in Fig. 2. OSS, Schirmer I test, CDEQ, FNIBUT and MNIBUT all exhibit robust discriminatory capacity with AUCs of 0.962, 0.953, 0.861, 0,799 and 0.769, respectively.
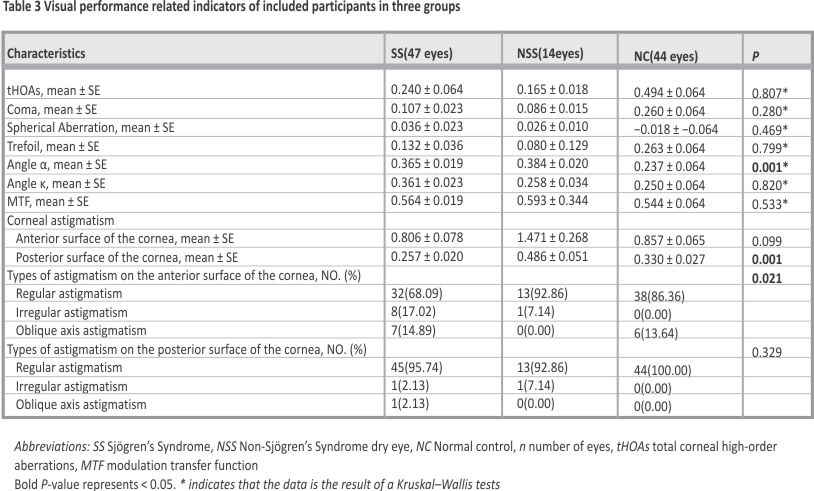
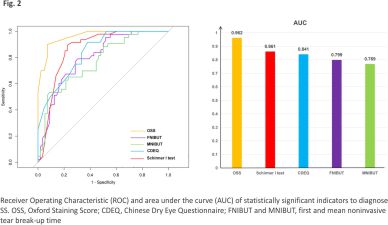 The results of the analysis of the indicators related to corneal optical quality are presented in Table 3. Angle α (p = 0.001), posterior corneal surface astigmatism (p = 0.001), and the types of astigmatism on the anterior (p = 0.0211) surface of the cornea were significantly different between the groups.
The results of the analysis of the indicators related to corneal optical quality are presented in Table 3. Angle α (p = 0.001), posterior corneal surface astigmatism (p = 0.001), and the types of astigmatism on the anterior (p = 0.0211) surface of the cornea were significantly different between the groups.
 The P-values in Tables 1, 2 and 3 represented the statistical significance of differences between the three groups (SSDE, NSSDE, and normal controls) for each variable. These values indicated whether there were significant differences in ocular surface manifestations and corneal optical quality indicators among the groups.
The P-values in Tables 1, 2 and 3 represented the statistical significance of differences between the three groups (SSDE, NSSDE, and normal controls) for each variable. These values indicated whether there were significant differences in ocular surface manifestations and corneal optical quality indicators among the groups.
Correlation of ocular surface manifestations and visual performance
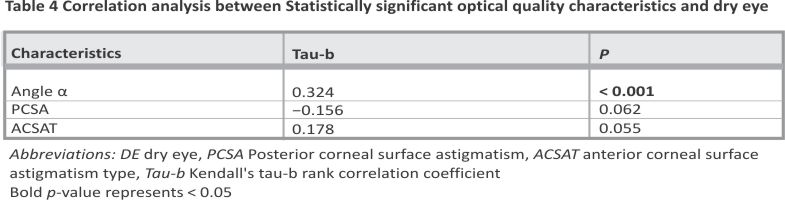
Correlation analyses were performed to assess the relationship between ocular surface manifestations and corneal optical quality indicators. Significant correlations identified in this step were further explored using multiple linear regression and multinomial logistic regression analyses to quantify the effect size and adjust for potential confounders (Table 4). Results confirmed a statistically robust association between Angle α and dry eye (DE) (Tau-b = 0.324, p < 0.001), while other parameters showed no significant correlations. We used multinomial linear regression analysis to investigate the relationship between DE and Angle α. All ocular surface indicators demonstrating statistically significant differences were incorporated as confounding factors in the regression model construction to control for potential bias.
 The groups of patients were not age-matched, so age and BCVA were treated as confounders in model 1. In Model 2, OSS, FNIBUT, MNIBUT, Schirmer I test, lipid layer distribution, and upper and lower MGL were also considered as confounders. The β-values of these models and their 95% confidence intervals, as well as the p-values, are presented in Table 5. The results showed that DE had a correlation with angle α, and this correlation was more pronounced in patients with angle α ≤ 0.3 (β = 0.062, 95% CI: 0.016,0.108; P = 0.01) than in patients with angle α > 0.3 (β = 0.048, 95% CI: 0.034,0.941; P = 0.035). This linear correlation remained in Model I, but was no longer statistically significant in Model 2.
The groups of patients were not age-matched, so age and BCVA were treated as confounders in model 1. In Model 2, OSS, FNIBUT, MNIBUT, Schirmer I test, lipid layer distribution, and upper and lower MGL were also considered as confounders. The β-values of these models and their 95% confidence intervals, as well as the p-values, are presented in Table 5. The results showed that DE had a correlation with angle α, and this correlation was more pronounced in patients with angle α ≤ 0.3 (β = 0.062, 95% CI: 0.016,0.108; P = 0.01) than in patients with angle α > 0.3 (β = 0.048, 95% CI: 0.034,0.941; P = 0.035). This linear correlation remained in Model I, but was no longer statistically significant in Model 2.
DE is now a major social and personal economic burden in some developed countries 22. According to epidemiological surveys, it is more prevalent in Asia than in Europe and North America 23. Women are the majority of patients with this disease, and the risk of the disease increases with age 24. In addition, female patients tend to be diagnosed at a younger age and have more severe symptoms compared to men 25.
As an autoimmune disease that affects multiple organ systems 26, SS is also more predominant in females 27,28. Studies have shown that SS causes chronic inflammation of the exocrine glands, which results in tissue destruction and dryness. Consequently, SS patients experience a decrease in aqueous tear production or secretion and are at a greater risk of developing DE.
SSDE can affect the tear film and ocular surface. Previously, researchers have compared dry eye performance between SS patients and healthy individuals. Numerous clinical studies have demonstrated ocular surface changes in SS patients in some classical clinical indicators, including decreased tear production, decreased tear break-up times (TBUT), and hyperosmolarity of tears 29,30. A study by J. Shimazaki et al. 31 stated that the lower eyelids of SS patients exhibited more pronounced MGL than other patients. All of these findings are in accordance with our results. Previous studies have shown that an AUC of 0.7–0.8 indicates that the indicator has acceptable discriminatory power 32. In this study, we have found that the Schirmer I test, OSS, CDEQ, FNIBUT and MNIBUT all exhibited robust discriminatory capacity, which was beneficial to distinguish patients with SS.
Destruction of the tear film and ocular surface can impair the corneal optical quality, which is the main cause of vision loss in patients with DE. In order to achieve a clear vision, it is important to maintain the integrity and stability of the precorneal tear film 33,34. In patients with DE, optical aberrations resulting from decreased tear stability and increased tear film breakup may negatively affect the observed image objectively and psychologically. Our study illustrated that indicators related to optical quality, such as angle α, showed statistical differences in SSDE patients compared to healthy controls. Astigmatism is a common symptom of DE 35. Furthermore, DE patients have higher tHOA as a consequence of the increased irregularities in their tear film 36. In the current study, the iTrace is the only ophthalmic instrument capable of measuring both the angles α and κ, enabling the assessment of visual quality in patients with DE disease. Post-operative follow-up results of some ophthalmic surgeries have shown that excessively large angles α can lead to deviation of the visual axis center, reducing the patient’s visual quality 37,38. Our findings highlight the significant impact of SSDE on corneal optical quality and ocular surface health. The identified correlations between ocular surface parameters and corneal optical quality suggest potential biomarkers for early intervention. This study demonstrates that SSDE significantly impairs corneal optical quality and ocular surface health. Early detection and management of SSDE are crucial to preserving visual quality.
In the present study, some patients completed questionnaires. The ocular surface disease index questionnaire (OSDI) has been employed as a diagnostic and grading tool for DE in numerous studies 39,40,41. However, this indicator was not statistically different between the two groups; this may be due to the small number of participants who completed the OSDI survey in this study. The Chinese Dry Eye Questionnaire (CDEQ) is designed for the characteristics of the living and working environments of Chinese, which makes it more suitable for Chinese patients with DE 42. Combined with our findings, this questionnaire may be more accurate than the OSDI in assessing the ocular surface of Chinese patients. The goal of this paper is to examine corneal surface symptoms and measurements in a Chinese SSDE group compared to non-SSDE patients and normal controls, and then to hopefully identify specific corneal surface symptoms and indicators that are more helpful in predicting optical quality. The strengths of our study are the inclusion of female patients with SSDE at a certain age and the wide variety of clinical indicators involved. These will greatly benefit patients with SS in terms of protecting their visual quality. This study identified significant correlations between ocular surface manifestations and corneal optical quality in Chinese female patients with SSDE. The findings highlight the importance of early detection and management of dry eye symptoms to preserve visual quality. Future research should explore the underlying mechanisms and potential therapeutic interventions to improve outcomes for patients with SSDE.
In spite of this, there are some limitations to this study. First, some participants provided incomplete or omitted information, which may have affected study results. In addition, the imbalance in the number of participants included in each group may cause some bias in the results. Second, although we have adjusted for the indicators, we have not completely eliminated the effects of unmeasured confounders. Another issue is that the number of cases in the groups of participants we included, as well as their ages, were not matched. Last but not least, this was a cross-sectional study. There is still a need for prospective and experimental studies to determine the causal relationship between visual performance and clinical ocular surface performance in patients with SS.
Conclusion
In conclusion, this study underscores the substantial impact of Sjogren’s syndrome dry eye (SSDE) on corneal optical quality and ocular surface health. Our findings highlight the critical importance of early detection and proactive management of SSDE in order to effectively preserve visual quality and mitigate potential long-term complications.
Data availability
The datasets used and/or analyzed in this study are available upon request from the corresponding author.
References
1. Stapleton F, Alves M, Bunya VY, Jalbert I, Lekhanont K, Malet F, Na K, Schaumberg D, Uchino M, Vehof J, et al. TFOS DEWS II Epidemiology Report. Ocul Surf. 2017;15(3):334–65.
2. Sheppard J, Shen Lee B, Periman LM. Dry eye disease: identification and therapeutic strategies for primary care clinicians and clinical specialists. ANN MED. 2023;55(1):241–52.
3. Akpek EK, Wirta DL, Downing JE, Tauber J, Sheppard JD, Ciolino JB, Meides AS, Krösser S. Efficacy and Safety of a Water-Free Topical Cyclosporine, 0.1%, Solution for the Treatment of Moderate to Severe Dry Eye Disease: The ESSENCE-2 Randomized Clinical Trial. Jama Ophthalmol. 2023;141(5):459 –66.
4. Nichols KK, Nichols JJ, Mitchell GL. The lack of association between signs and symptoms in patients with dry eye disease. Cornea. 2004;23(8):762–70.
5. Yang Y, Zhong J, Cui D, Jensen LD. Up-to-date molecular medicine strategies for management of ocular surface neovascularization. ADV DRUG DELIVER REV. 2023;201: 115084.
6. Tsubota K, Yokoi N, Shimazaki J, Watanabe H, Dogru M,
Yamada M, Kinoshita S, Kim H, Tchah H, Hyon JY, et al. New Perspectives on Dry Eye Definition and Diagnosis: A Consensus Report by the Asia Dry Eye Society. Ocul Surf. 2017;15(1):65–76.
7. Yu K, Bunya V, Maguire M, Asbell P, Ying G. Systemic Conditions Associated with Severity of Dry Eye Signs and Symptoms in the Dry Eye Assessment and Management Study. Ophthalmology. 2021;128(10):1384–92.
8. He J, Chen J, Miao M, Zhang R, Cheng G, Wang Y, Feng R, Huang B, Luan H, Jia Y, et al. Efficacy and Safety of Low-Dose Interleukin 2 for Primary Sjögren Syndrome: A Randomized Clinical Trial. JAMA NETW OPEN. 2022;5(11): e2241451.
9. Baer AN, Gottenberg J, St Clair EW, Sumida T, Takeuchi T, Seror R, Foulks G, Nys M, Mukherjee S, Wong R, et al. Efficacy and safety of abatacept in active primary Sjögren’s syndrome: results of a phase III, randomised, placebo-controlled trial. In. 2021;80: 339–48.
10. Brito-Zerón P, Theander E, Baldini C, Seror R, Retamozo S, Quartuccio L, Bootsma H, Bowman SJ, Dörner T, Gottenberg J, et al. Early diagnosis of primary Sjögren’s syndrome: EULAR-SS task force clinical recommendations. EXPERT REV CLIN IMMU. 2016;12(2): 137–56.
11. Bjordal O, Norheim KB, Rødahl E, Jonsson R, Omdal R. Primary Sjögren’s syndrome and the eye. SURV OPHTHALMOL. 2020;65(2):119–32.
12. Baudouin C, Aragona P, Messmer EM, Tomlinson A, Calonge M, Boboridis KG, Akova YA, Geerling G, Labetoulle M, Rolando M: Role of Hyperosmolarity in the Pathogenesis and Management of Dry Eye Disease: Proceedings of the OCEAN Group Meeting. The Ocular Surface. 2013;11(4):246–258.
13. Baudouin C, Irkeç M, Messmer EM, Benítez-Del-Castillo JM, Bonini S, Figueiredo FC, Geerling G, Labetoulle M, Lemp M, Rolando M, et al. Clinical impact of inflammation in dry eye disease: proceedings of the ODISSEY group meeting. ACTA OPHTHALMOL. 2018;96(2):111–9.
14. Akpek EK, Bunya VY, Saldanha IJ. Sjögren’s Syndrome: More Than Just Dry Eye. Cornea. 2019;38(5):658–61.
15. Franceschini F, Cavazzana I, Andreoli L, Tincani A. The 2016 classification criteria for primary Sjogren’s syndrome: what’s new? BMC MED. 2017;15(1):69.
16. J. W: Ocular Surface Disease Index (OSDI) Administration and Scoring Manual. In: Irvine, CA: Allergan, Inc.; 2004.
17. Zhao H, Liu Z, Yang W, Xiao X, Chen J, Li Q, Zhong T: [Development and assessment of a dry eye questionnaire applicable to the Chinese population]. [Zhonghua yan ke za zhi] Chinese journal of ophthalmology. 2015;51 (9):647–654.
18. Wang MTM, Craig JP. Comparative Evaluation of Clinical Methods of Tear Film Stability Assessment: A Randomized Crossover Trial. JAMA OPHTHALMOL. 2018;136(3):291–4.
19. Begley C, Caffery B, Chalmers R, Situ P, Simpson T, Nelson JD. Review and analysis of grading scales for ocular surface staining. Ocul Surf. 2019;17 (2):208–20.
20. Tashbayev B, Yazdani M, Arita R, Fineide F, Utheim TP. Intense pulsed light treatment in meibomian gland dysfunction: A concise review. Ocul Surf. 2020;18(4):583–94.
21. Eom Y, Lee J, Keun Lee H, Myung Kim H, Suk Song J: Comparison of conjunctival staining between lissamine green and yellow filtered fluorescein sodium. Canadian journal of ophthalmology. Journal canadien d’ophtalmologie. 2015;50(4):273– 277.
22. McKnight W: Chapter Ten – Operational Big Data: Key-Value, Document, and Column Stores: Hash Tables Reborn. In: Information Management. Edited by McKnight W. Boston: Morgan Kaufmann; 2014: 97–109.
23. Tsubota K, Pflugfelder SC, Liu Z, Baudouin C, Kim HM, Messmer EM, Kruse F, Liang L, Carreno-Galeano JT, Rolando M, et al. Defining Dry Eye from a Clinical Perspective. Int J Mol Sci. 2020;21(23):9271.
24. Messmer EM: The pathophysiology, diagnosis, and treatment of dry eye disease. DTSCH ARZTEBL INT 2015, 112(5):71–81, 82.
25. Matossian C, McDonald M, Donaldson KE, Nichols KK, MacIver S, Gupta PK: Dry Eye Disease: Considerations for Women’s Health. Journal of women’s health (2002) 2019, 28(4):502–514.
26. Chen W, Cao H, Lin J, Olsen N, Zheng SG. Biomarkers for Primary Sjögren’s Syndrome. Genomics, Proteomics, Bioinformatics. 2015;13(4):219–23.
27. Manuel RSJ, Liang Y. Sexual dimorphism in immunometabolism and autoimmunity: Impact on personalized medicine. AUTOIMMUN REV. 2021; 20(4): 102775.
28. Brandt JE, Priori R, Valesini G, Fairweather D. Sex differences in Sjögren’s syndrome: a comprehensive review of immune mechanisms. BIOL SEX DIFFER. 2015;6:19.
29. Stevenson W, Chauhan SK, Dana R: Dry eye disease: an immune-mediated ocular surface disorder. Archives of ophthalmology (Chicago, Ill.: 1960) 2012, 130(1):90–100.
30. Lemp MA. Advances in Understanding and Managing Dry Eye Disease. Am J Ophthalmol. 2008;146(3):350–6.
31. Shimazaki J, Goto E, Ono M, Shimmura S, Tsubota K. Meibomian gland dysfunction in patients with Sjögren syndrome. Ophthalmology. 1998;105 (8):1485–8.
32. Mandrekar JN. Receiver operating characteristic curve in diagnostic test assessment. Journal of thoracic oncology: official publication of the International Association for the Study of Lung Cancer. 2010;5(9):13 15–6.
33. Rieger G. The importance of the precorneal tear film for the quality of optical imaging. Br J Ophthalmol. 1992; 76(3):157–8.
34. Tutt R, Bradley A, Begley C, Thibos LN. Optical and visual impact of tear break-up in human eyes. Invest Ophth Vis Sci. 2000;41(13):4117–23.
35. Liu Z, Pflugfelder SC. Corneal surface regularity and the effect of artificial tears in aqueous tear deficiency. Ophthalmology. 1999;106(5):939–43.
36. Montés-Micó R, Cáliz A, Alió JL: Wavefront analysis of higher order aberrations in dry eye patients. Journal of refractive surgery (Thorofare, N.J.: 1995) 2004, 20(3):243–247.
37. Fu Y, Kou J, Chen D, Wang D, Zhao Y, Hu M, Lin X, Dai Q, Li J, Zhao Y. Influence of angle kappa and angle alpha on visual quality after implantation of multifocal intraocular lenses. J Cataract Refr Surg. 2019;45(9):1258– 64.
38. Wang L. Guimaraes de Souza R, Weikert MP, Koch DD: Evaluation of crystalline lens and intraocular lens tilt using a swept-source optical coherence tomography biometer. J Cataract Refr Surg. 2019;45(1):35–40.
39. de Paiva CS, Trujillo-Vargas CM, Schaefer L, Yu Z, Britton RA, Pflugfelder SC. Differentially Expressed Gene Pathways in the Conjunctiva of Sjögren Syndrome Keratoconjunctivitis Sicca. Front Immunol. 2021;12: 702755.
40. Ren X, Chou Y, Wang Y, Jing D, Chen Y, Li X. The Utility of Oral Vitamin B1 and Mecobalamin to Improve Corneal Nerves in Dry Eye Disease: An In Vivo Confocal Microscopy Study. Nutrients. 2022;14(18):3750.
41. Tsai T, Alwees M, Rost A, Theile J, Dick HB, Joachim SC, Taneri S. Changes of Subjective Symptoms and Tear Film Biomarkers following Femto-LASIK. Int J Mol Sci. 2022;23(14):7512.
42. Zhao Hui LMAY. Development and evaluation of a dry eye questionnaire in China. Chinese J Ophth. 2015;9: 647–54.
Acknowledgements
Support for this work was provided by the Guangzhou Science and Technology Project, Natural Science Foundation Project of Guangdong Province, and Guangzhou Health Science and Technology Project.
Funding
This study was supported by Guangzhou Science and Technology Project (No. 2024A03J0207), Natural Science Foundation Project of Guangdong Province (No. 2019A1515011094), and Guangzhou Health Science and Technology Project (No. 205010606044).
Author information
Author notes
Jingyu Zhang, Qian Deng and Maierhaba Maitiyaer contributed equally and should be considered as co-first authors.
Authors and Affiliations
Ophthalmic Center, The Second Affiliated Hospital, Guangzhou Medical University, Guangzhou, Guangdong, China
Jingyu Zhang, Qian Deng, Li Wang, Yue Liang, Qiudan Huang & Zhiping Liu
Department of Rheumatology, The Second Affiliated Hospital, Guangzhou Medical University, Guangzhou, Guangdong, China
Maierhaba Maitiyaer & ShuiLian Yu
Guangdong Provincial Key Laboratory of Allergy & Clinical Immunology, The Second Affiliated Hospital, Guangzhou Medical University, Guangzhou, Guangdong, China
Jingyu Zhang, Maierhaba Maitiyaer, Li Wang, Yue Liang, ShuiLian Yu & Zhiping Liu
Department of Ophthalmology, Zhejiang Provincial People’s Hospital, Bijie Hospital, Bijie, Guizhou, China
Qian Deng
Department of Ophthalmology, University of Colorado, Aurora, CO, USA
Amy Michelle Huang
Contributions
Author Contributions: JZ and ZL designed the study; QD, MM, LW, YL, QH, and SY participated in data collection, analysis and interpretation; JZ, AH, and ZL drafted the manuscript; All authors reviewed and approved the manuscript.
Corresponding authors
Correspondence to ShuiLian Yu or Zhiping Liu.
Credit: Zhang, J., Deng, Q., Maitiyaer, M. et al. The relation between corneal optical quality and clinical ocular surface manifestations in Chinese females with Sjogren’s syndrome dry eye. BMC Ophthalmol 25, 399 (2025). https://doi.org/ 10.1186/s12886-025-04225-2