Wufei Chen, Pan Gao, Fang Lu, Ernuo Wang, Haiquan Liu & Ming Li
Abstract
Background
Mutations in the human receptor tyrosine kinase epidermal growth factor receptor-2 (HER2) are rare. This study aimed to investigate the clinical characteristics and computed tomography (CT) texture features of lung adenocarcinoma (LUAD) patients with HER2 mutation.
Methods
This study included 933 LUAD patients from January 2018 to December 2023 and classified their CT textures accordingly.
Results
The data indicated that the incidence of HER2 mutation was higher in younger LUAD patients than in older patients [7.5% (31/413) vs. 1.5% (8/520), p < 0.0001] and was associated with never-smokers [0% (0/78) vs. 4.6% (39/855), p = 0.03]. In this study, the tumors were categorized based on their diameter into T1a and ≥T1b. The data revealed that HER2 mutation was more frequent in T1a than in ≥T1b [11.0% (23/210) vs. 2.2% (16/723), p < 0.0001]. Furthermore, non-pSD was more common than pSD in LUAD with HER2 mutation than in LUAD with HER2 wild type [82.1% (31/39) vs.17.9% (7/39), p = 0.01]. Moreover, the size of pGGO (0.94 ± 0.29 cm vs. 1.24 ± 0.39 cm, p = 0.0009), mGGO (0.86 ± 0.39 cm vs. 1.5 ± 0.77 cm, p < 0.0001) and pSD (1.75 ± 0.81 cm vs. 2.5 ± 1.4 cm, p < 0.05) in LUAD patients with HER2 mutation was smaller than those with HER2 wild-type patients. In addition, when LUADs with HER2 wild type transformed from pGGO to mGGO, their sizes increased significantly (1.50 ± 0.77 cm vs. 1.24 ± 0.39 cm). It was also observed that the incidence of LUAD with HER2 mutation of ≤1 cm was significantly more than that of > 1 cm compared to that in LUAD with HER2 wild type [14.8% (12/81) vs. 1.7% (3/173), p < 0.0001].
Conclusion
This study indicated that the incidence of HER2 mutation was higher in younger and never-smoking LUAD patients. Furthermore, the growth of LUAD with HER2 mutation was slower than that of those with HER2 wild type. Moreover, most LUAD with HER2 mutation changed into pSD after > 1 cm.
Keywords: Lung adenocarcinoma, HER2 mutation, Smoking, pGGO, mGGO
Introduction
Lung cancer is a leading cause of cancer-related death globally. The World Health Organization estimated that in 2020, lung cancer caused 1.76 million deaths, accounting for approximately 19% of the total cancer-related deaths 1,2. Non-small cell lung cancer (NSCLC) comprises 80 to 85% of all lung cancers, and 40% of these are adenocarcinoma histologically 3. In 2004, it was identified that epidermal growth factor receptor gene (EGFR) mutation was associated with lung adenocarcinoma (LUAD) patients 4,5, and advancements in genomic sequencing and targeted therapy have further sub-classified LUAD based on the driver oncogenes.
Human epidermal growth factor 2 (HER2, also known as ERBB2, NEU, or EGFR2) is a member of the ERBB family of receptor tyrosine kinases. It is encoded by the ERBB2 gene, which is an important driver oncogene for lung cancer. The ERBB2 gene is located on the long arm of chromosome 17 (17 q21) and has been observed to activate the downstream signalling pathways, such as PI3K-AKT and MEK-ERK, which cause cell proliferation and migration 6,7. Several studies have indicated that HER2 mutations in exon 20 cause constitutive activation of downstream signalling and promote the development of lung tumors in mouse models 8. Moreover, proteins encoded by the HER2 gene are a tyrosine kinase receptor of the ErbB family. In addition, it has been observed that HER2 has no direct activating ligand and serves as the preferred and most stable heterodimerization partner for all the other family receptors, especially EGFR 9.
The most economical and effective non-invasive technique to diagnose lung cancer is a Computed Tomography (CT) scan 10. Furthermore, it is significant and interesting to understand the imaging features of some special lung cancers, and it is potentially useful for guiding lung cancer therapy.
Radiogenomics is a field that investigates the radiological appearance of tumors as well as the genomic alterations, such as driver oncogenes 11. However, studies on the imaging features of LUAD with HER2 mutation are limited. Therefore, this study aimed to assess the clinical characteristics and CT texture features of LUAD patients with HER2 mutation.
Methods
Patients
This study followed the Declaration of Helsinki and was authorized by the Ethics Review Board of Huadong Hospital, affiliated with Fudan University (approval number 2023 0108). Because of its retrospective nature, the requirement of patient-informed consent was waived for this study. A total of 933 histologically confirmed LUAD patients with primary lung cancer from January 2018 to December 2023 were included in this study. All tissue specimens were obtained surgically, and no pre-operative treatments were given. Patients were selected consecutively. The inclusion criteria included patients who (a) were pathologically diagnosed with LUAD, (b) had HER2 mutation test reports, and (c) had clinical data, including age, sex, and smoking history. All 933 patients met the inclusion criteria, and of these, only 818 patients had preoperative CT scans, which were used to evaluate the CT features of the lesion.
Computed tomographic assessment
For preoperative chest CT scans, three scanners, including GE Discovery CT750 HD, 64-slice Light Speed VCT (GE Medical Systems), and Somatom Definition Flash, were employed. The scanning parameters were: 120 kVp, 100–200 mAs, 0.75–1.5 pitch range, and 1–1.25 mm collimation widths. Furthermore, for all the imaging data, a medium sharp reconstruction algorithm was used, which provided 1–1.25 mm thick images.
Interpretation of computed tomographic images
The CT images were reviewed by 2 radiologists with 10 to 12 years of experience in chest CT diagnosis independently. The radiologists were not blinded to the surgical resection of LUAD patients; however, they were blinded to clinical data as well as gene expression and mutational status. For CT image analysis, both mediastinal (width = 350 HU; level = 40 HU) and lung (width = 1500 HU; level = -650 HU) window settings were employed. Moreover, lesion location, size, and texture were retrospectively analyzed. In addition, the lesions’ long-axis diameter at their largest section was also measured. Furthermore, lesions with ground-glass opacities were categorized as pure GGO (pGGO), those with 1% < GGO < 100% were classified as mixed GGO (mGGO), while those lacking any GGO were deemed pure solid LUADs (pSD). For statistical analysis, pGGO and mGGO categories were combined as non-pSD. In the case of multiple lesions on images, the largest lesion was selected for observation.
Statistical analysis
SAS version 9.4 (SAS Institute Inc., Cary, NC) was employed for all the statistical analyses. The distribution differences in categorical variables were assessed via the chi-square (χ2) test, whereas an independent t-test was performed to assess differences in continuous variables. The p-value < 0.05 was deemed statistically significant.
Results
Patient demographics characteristics
This study included a total of 933 LUAD patients aged between 18 and 86 years (average age = 61.8 years), including 393 females and 540 males. Of these 933 patients, 39 (4.2%) had an HER2 mutation, and 78 patients had a history of smoking.
Correlation of HER2 status and clinical factors
Statistical analysis (Table 1) indicated that the incidence of HER2 mutation was higher in younger LUAD patients than in older patients [7.5% (31/ 413) vs. 1.5% (8/520), p < 0.0001]. Furthermore, correlation analysis between HER2 and smoking revealed that HER2 mutation was negatively associated with smoking in the whole cohort [0% (0/78) vs. 4.6% (39/855), p = 0.03].
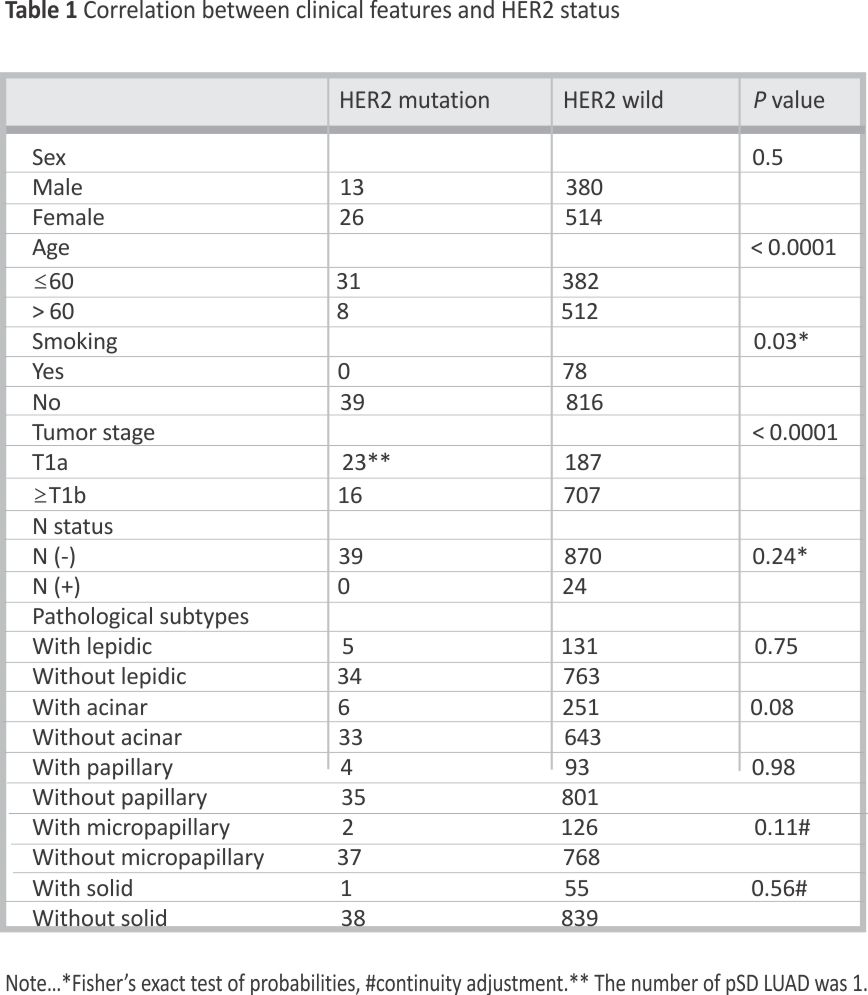 Based on their diameter, the tumors were categorized into ≤1 cm and > 1 cm. The results indicated that HER2 mutation was more frequent in small tumors than in larger ones [11.0% (23/210) vs. 2.2% (16/723), p < 0.0001]. Lymph node metastasis and HER2 mutations were correlated. The tumor was also categorized into histopathological subtypes with or without some components, and there was no statistically significant correlation between HER2 mutation and histopathological subtypes.
Based on their diameter, the tumors were categorized into ≤1 cm and > 1 cm. The results indicated that HER2 mutation was more frequent in small tumors than in larger ones [11.0% (23/210) vs. 2.2% (16/723), p < 0.0001]. Lymph node metastasis and HER2 mutations were correlated. The tumor was also categorized into histopathological subtypes with or without some components, and there was no statistically significant correlation between HER2 mutation and histopathological subtypes.
Computed tomographic texture of HER2 mutation and wild-type LUAD patients
Of the 933 patients, 818 patients had CT scan data, and 38 were positive for HER2 mutation. Of these 818 patients with CT scans, 754 had single lesions, while 64 had multiple lesions. The data showed that non-pSD was more common than pSD in LUAD with HER2 mutation than in LUAD with HER2 wild type [82.1% (31/39) vs.17.9% (7/39), p = 0.01] (Table 2).
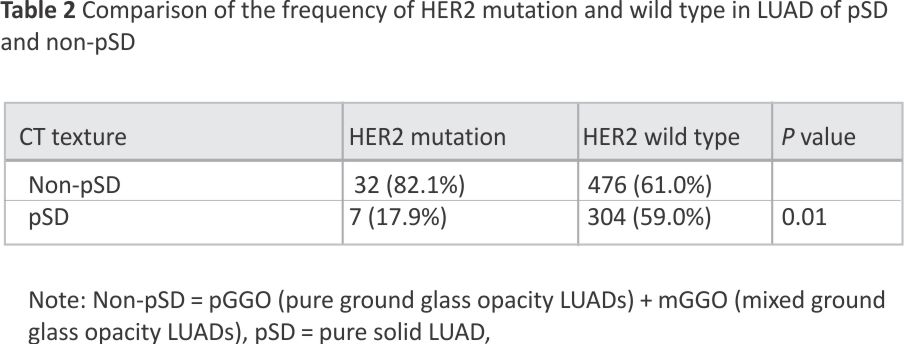 Comparison between LUADs of pGGO and mGGO in HER2 mutation and wild-type LUAD patients
Comparison between LUADs of pGGO and mGGO in HER2 mutation and wild-type LUAD patients
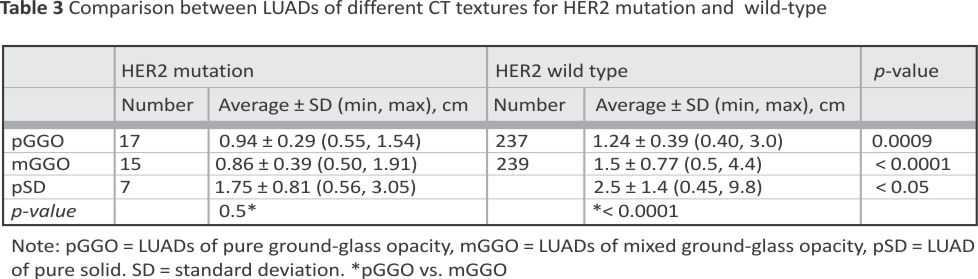
The CT data from three textures indicated that the size of pGGO (0.94 ± 0.29 cm vs. 1.24 ± 0.39 cm, p = 0.0009), mGGO (0.86 ± 0.39 cm vs. 1.5 ± 0.77 cm, p < 0.0001) and pSD (1.75 ± 0.81 cm vs. 2.5 ± 1.4 cm, p < 0.05) in LUAD patients with HER2 mutation was smaller than those with HER2 wild-type patients. There was no size difference for HER2 mutation lesions between pGGO and mGGO. However, in LUAD with HER2 wild-type patients, mGGO lesions were significantly larger than pGGO (1.50 ± 0.77 cm vs. 1.24 ± 0.39 cm) (Table 3).
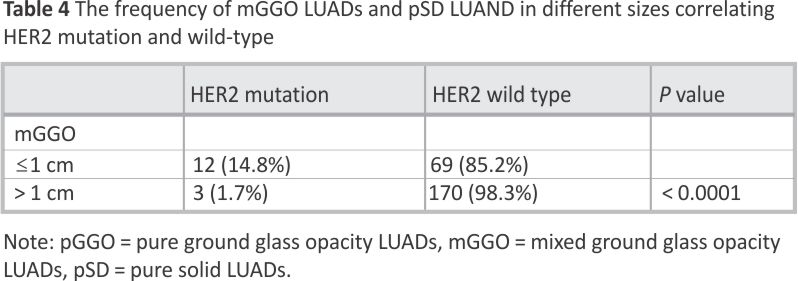
 Furthermore, LUAD lesions were categorized into ≤1 cm and > 1 cm, and the frequency of ≤1 cm LUAD lesions with HER2 mutation was significantly higher than that of > 1 cm compared to HER2 wild type [14.8% (12/81) vs. 1.7% (3/173), p < 0.0001] (Table 4).
Furthermore, LUAD lesions were categorized into ≤1 cm and > 1 cm, and the frequency of ≤1 cm LUAD lesions with HER2 mutation was significantly higher than that of > 1 cm compared to HER2 wild type [14.8% (12/81) vs. 1.7% (3/173), p < 0.0001] (Table 4).
The literature has estimated that the incidence of HER2 mutations is 2–6.7% in NSCLC adenocarcinomas 12,13,14,15,16,17, which is consistent with this study (4.2%). The association of HER2 mutation with smoking remains controversial. Some studies have suggested that HER2 mutation does not correlate with smoking 15,18,19. However, most studies have indicated that HER2 mutation is more common in non-smokers 16, 20,21,22, such as a study of 666 HER2 mutations in 13,920 patients 18. In this study, the incidence of smokers was 8.4%, which was significantly lower than 55.8% of that reported by H. Shigematsu et al. 16 in the Asian group. This discriment might be because in their study, the HER2 mutation was only 2.4%. Our study revealed that HER2 mutation was associated with non-smokers and was statistically significant, which was consistent with most studies.
Several studies correlate HER2 mutation with younger age patients, 19–21. However, this is still controversial as some studies suggest that HER2 mutations are not correlated with age 14,22. Furthermore, some literature suggests that HER2 mutations are associated with the female gender 16,23–26, while some deny it 15,19,22, 27. This study showed that HER2 mutation was more common in younger patients and women than in older individuals and men; however, there was no statistical difference, which might be because of the small sample size and warrants further research with a larger sample size.
This study indicated that LUAD with HER2 mutation was more frequent in tumors with T1a cm than those≥T1b, which was consistent with the data from the previous study. R. Zhao et al. 18 found LUAD with HER2 mutation to be more common in early LUAD and inversely proportional to the degree of invasion. However, they found LUAD with HER2 mutation frequently correlated with lepidic components, which is inconsistent with the present study. Here, correlation was observed between HER2 mutation and histopathological subtypes. However, further experimental studies are required to validate these findings.
Previous studies on CT imaging features in LUAD with HER2 mutation were scarce because HER2 mutations are rare in lung cancer. Here, a larger sample of LUAD CT characteristics was analyzed, which indicated that non-PSD was more common than pSD in HER2 mutation LUAD patients.
The genomic landscape analysis of 37 patients with ground glass opacities (GGOs) revealed that ERB2 had a higher mutation frequency, accounting for 8% 28. Another study showed that LUAD GGO tumors had a higher HER2 mutation incidence, 24, partially consistent with this study. X. Liu et al. 30 showed that LUAD with HER2 mutation was more frequently classified as cluster 4 lesion (small GGO lesions with a maximum diameter of 1.0 ± 0.9 cm). Here, the average sizes of HER2 mutation carrying LUAD pGGO and mGGO were 0.94 ± 0.29 cm and 0 0.86 ± 0.39 cm, respectively, which are in line with the results of X. Liu et al. 29.
The theory of progressive radiologic evolution from non-solid LUADs to part-solid and forming most solid LUADs is well known 30, 31. Studies have shown that a GGO LUAD initially increases in size, followed by the appearance and subsequent enlargement of a solid portion within the lesion 32. Furthermore, it has been observed that lesions that transition from pGGO to mGGO or from mGGO to solid have a rapid increase in size 33,34,35. Here, it was observed that the size of pGGO or mGGO in the HER2 wild-type LUAD was significantly larger than those with HER2 mutation LUAD. Moreover, when the LUADs transformed from pGGO into mGGO, the sizes of LUADs with HER2 wild type increased significantly, but that with HER2 mutation did not change significantly. In addition, for mGGO lesions with HER2 mutation, the frequency of ≤1 cm was more than that of > 1 cm compared to HER2 wild type. This suggested that most LUADs with HER2 mutation changed into pSD after 1 cm.
Overall, this study indicated that LUAD with HER2 mutation was smaller in size than LUAD with HER2 wild type for GGO-LUAD, mGGO-LUAD, or pSD-LUAD, respectively. Therefore, it can be inferred that LUAD with HER2 mutation had a slower growth speed than their counterpart. Furthermore, as LUAD with HER2 mutation transformed into mGGO, the size did not increase as observed in LUADs with HER2 wild type. This study will provide a reference for future research on this question.
Limitations
This study has certain limitations. First, the study sample was small; therefore, l, the details of HER mutation tumor stratification could not be analyzed. Second, the analysis was limited to adenocarcinoma, and other histologic subtypes were not addressed. However, the majority of HER2 mutations are found in adenocarcinoma. Third, no survival data were collected in this study; therefore, the relationship between tumors of different textures and survival was not assessed.
Conclusion
In summary, this study revealed that HER2 mutation was more common in younger and never-smoking LUAD patients. Furthermore, most LUADs manifested as non-pSD in CT scans. Moreover, the size of LUAD with HER2 mutation was smaller than that with HER2 wild type, and most LUAD with HER2 mutation transformed into pSD after their size increased to> 1 cm.
Data availability
All data generated or analysed during this study are included in this published article.
Abbreviations
HER2: Human epidermal growth factor 2
EGFR: Epidermal growth factor receptor gene
CT: Computed tomography
LUAD: Lung adenocarcinoma
pGGO: Pure ground glass opacity
mGGO: Mixed ground glass opacity
pSD: Pure solid
NSCLC: Non-small cell lung cancer
References
1. Thai AA, Solomon BJ, Sequist LV, Gainor JF, Heist RS. Lung cancer. Lancet (London, England). 2021; 398(10299): 535–54.
2. Zhang Y, Elgizouli M, Schöttker B, Holleczek B, Nieters A, Brenner H. Smoking-associated DNA methylation markers predict lung cancer incidence. Clin Epigenetics. 2016; 8:127.
3. Ladanyi M. W. P. Pao Lung adenocarcinoma: guiding EGFR-targeted therapy and beyond. Mod Pathology: Official J United States Can Acad Pathol Inc 21: 2008;2:S16–22.
4. Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat SM, Supko JG, Haluska FG, Louis DN, Christiani DC, Settleman J, Haber DA. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004;350( 21):2129–39.
5. Paez JG, Jänne PA, Lee JC, Tracy S, Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ, Naoki K, Sasaki H, Fujii Y, Eck MJ, Sellers WR, Johnson BE, Meyerson M. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Sci (New York N Y). 2004;304(5676):1497–500.
6. Xu H, Liang Q, Xu X, Tan S, Wang S, Liu Y, Liu L. Afatinib combined with anlotinib in the treatment of a lung adenocarcinoma patient with a novel HER2 mutation: a case report and review of the literature. World J Surg Oncol. 2021;19 (1):330.
7. Peters S, Zimmermann S. Targeted therapy in NSCLC driven by HER2 insertions. Translational lung cancer Res 3(2)2014;84– 8.
8. Pillai RN, Behera M, Berry LD, Rossi MR, Kris MG, Johnson BE, Bunn PA, Ramalingam SS, Khuri FR. HER2 mutations in lung adenocarcinomas: a report from the Lung Cancer Mutation Consortium. Cancer. 2017;123(21): 4099–105.
9. Sawan P, Plodkowski AJ, Li AE, Li BT, Drilon A, Capanu M, Ginsberg MS. CT features of HER2-mutant lung adenocarcinomas. Clin Imaging. 2018; 51:279–83.
10. Yue JY, Chen J, Zhou FM, Hu Y, Li MX, Wu QW, Han DM. CT-pathologic correlation in lung adenocarcinoma and squamous cell carcinoma. Medicine. 2018;97 (50):e13362.
11. Mazurowski MA. Radiogenomics: what it is and why it is important. J Am Coll Radiology: JACR. 2015;12(8):862–6.
12. Garrido-Castro AC, Felip E. HER2-driven non-small cell lung cancer (NSCLC): potential therapeutic approaches. Translational Lung Cancer Res. 2013;2 (2):122–7.
13. Kim EK, Kim KA, Lee CY, Shim HS. The frequency and clinical impact of HER2 alterations in lung adenocarcinoma. PLoS ONE. 2017;12(2):e0171280.
14. Li C, Fang R, Sun Y, Han X, Li F, Gao B, Iafrate AJ, Liu XY, Pao W, Chen H, Ji H. Spectrum of oncogenic driver mutations in lung adenocarcinomas from East Asian never smokers. PLoS ONE. 2011;6(11):e28204.
15. Li X, Zhao C, Su C, Ren S, Chen X, Zhou C. Epidemiological study of HER-2 mutations among EGFR wild-type lung adenocarcinoma patients in China. BMC Cancer. 2016;16(1):828.
16. Shigematsu H, Takahashi T, Nomura M, Majmudar K, Suzuki M, Lee H, Wistuba II, Fong KM, Toyooka S, Shimizu N, Fujisawa T, Minna JD, Gazdar AF. Somatic mutations of the HER2 kinase domain in lung adenocarcinomas. Cancer Res. 2005;65 (5):1642–6.
17. Tan AC, Saw SPL, Chen J, Lai GGY, Oo HN, Takano A, Lau DPX, Yeong JPS, Tan GS, Lim KH, Skanderup AJ, Chan JWK, Teh YL, Rajasekaran T, Jain A, Tan WL, Ng QS, Kanesvaran R, Lim WT, Ang MK, Tan DSW. Clinical and genomic features of HER2 exon 20 insertion mutations and characterization of HER2 expression by immunohistochemistry in East Asian Non-small-cell Lung Cancer. JCO Precision Oncol. 2022; 6:e2200278.
18. Zhao R, Li J, Guo L, Xiang C, Chen S, Zhao J, Shao J, Zhu L, Ye M, Qin G, Chu T, Han Y. EGFR and ERBB2 exon 20 insertion mutations in Chinese non-small cell Lung Cancer patients: pathological and molecular characterization, and first-line systemic treatment evaluation. Target Oncol. 2024;19(2):277–88.
19. Arcila ME, Chaft JE, Nafa K, Roy-Chowdhuri S, Lau C, Zaidinski M, Paik PK, Zakowski MF, Kris MG, Ladanyi M. Prevalence, clinicopathologic associations, and molecular spectrum of ERBB2 (HER2) tyrosine kinase mutations in lung adenocarcinomas. Clin Cancer Research: Official J Am Association Cancer Res. 2012;18(18):4910–8.
20. Zhao M, Zhan C, Li M, Yang X, Yang X, Zhang Y, Lin M, Xia Y, Feng M, Wang Q. Aberrant status and clinicopathologic characteristic associations of 11 target genes in 1,321 Chinese patients with lung adenocarcinoma. J Thorac Disease. 2018;10(1):398–407.
21. Wu X, Zhao J, Yang L, Nie X, Wang Z, Zhang P, Li C, Hu X, Tang M, Yi Y, Du X, Xia X, Guan Y, Yu Z, Gu W, Quan X, Li L, Shi H. Next-generation sequencing reveals Age-dependent genetic underpinnings in lung adenocarcinoma. J Cancer. 2022;13(5):1565–72.
22. Hsu KH, Ho CC, Hsia TC, Tseng JS, Su KY, Wu MF, Chiu KL, Yang TY, Chen KC, Ooi H, Wu TC, Chen HJ, Chen HY, Chang CS, Hsu CP, Hsia JY, Chuang CY, Lin CH, Chen JJ, Chen KY, Liao WY, Shih JY, Yu SL, Yu CJ, Yang PC, Chang GC. Identification of five driver gene mutations in patients with treatment-naïve lung adenocarcinoma in Taiwan. PLoS ONE. 2015;10(3):e01 20852.
23. Mazières J, Peters S, Lepage B, Cortot AB, Barlesi F, Beau-Faller M, Besse B, Blons H, Mansuet-Lupo A, Urban T, Moro-Sibilot D, Dansin E, Chouaid C, Wislez M, Diebold J, Felip E, Rouquette I, Milia JD, Gautschi O. Lung cancer that harbors an HER2 mutation: epidemiologic characteristics and therapeutic perspectives. J Clin Oncology: Official J Am Soc Clin Oncol. 2013;31(16):1997–2003.
24. Eng J, Hsu M, Chaft JE, Kris MG, Arcila ME, Li BT. Outcomes of chemotherapies and HER2-directed therapies in advanced HER2-mutant lung cancers, lung cancer (Amsterdam, Netherlands). 2016;99:53–6.
25. Pao W, Girard N. New driver mutations in non-small-cell lung cancer, The Lancet. Oncology. 2011;12(2):175– 80.
26. Li C., Sun Y., Fang R., Han X., Luo X., Wang R., Pan Y., Hu H., Zhang Y., Pao W., Shen L., Ji H., Chen H. Lung adenocarcinomas with HER2-activating mutations are associated with distinct clinical features and HER2/EGFR copy number gains. J Thorac Oncology: Official Publication Int Association Study Lung Cancer. 2012;7(1): 85–9.
27. Wu D, Xie Y, Jin C, Qiu J, Hou T, Du H, Chen S, Xiang J, Shi X, Liu J. The landscape of kinase domain duplication in Chinese lung cancer patients. Annals Translational Med. 2020;8(24):1642.
28. Cao P, Hu S, Kong K, Han P, Yue J, Deng Y, Zhao B, Li F. Genomic landscape of ground glass opacities (GGOs) in East asians. J Thorac Disease. 2021;13(4): 2393–403.
29. Liu X, Xu T, Wang S, Chen Y, Jiang C, Xu W, Gong J. CT-based radiomic phenotypes of lung adenocarcinoma: a preliminary comparative analysis with targeted next-generation sequencing. Front Med. 2023;10: 1191019.
30. Jung W, Cho S, Yum S, Chung JH, Lee KW, Kim K, Lee CT, Jheon S. Stepwise Disease Progression Model of Subsolid Lung Adenocarcinoma with cystic airspaces. Ann Surg Oncol. 2020;27 (11):4394–403.
31. Kakinuma R, Noguchi M, Ashizawa K, Kuriyama K, Maeshima AM, Koizumi N, Kondo T, Matsuguma H, Nitta N, Ohmatsu H, Okami J, Suehisa H, Yamaji T, Kodama K, Mori K, Yamada K, Matsuno Y, Murayama S, Murata K. Natural history of Pulmonary Subsolid nodules: a prospective Multicenter Study. J Thorac Oncology: Official Publication Int Association Study Lung Cancer. 2016;11(7):1012–28.
32. Takashima S, Maruyama Y, Hasegawa M, Yamanda T, Honda T, Kadoya M, Sone S. CT findings and progression of small peripheral lung neoplasms having a replacement growth pattern, AJR. Am J Roentgenol. 2003;180(3):817–26.
33. Lee SW, Leem CS, Kim TJ, Lee KW, Chung JH, Jheon S, Lee JH, Lee CT. The long-term course of ground-glass opacities detected on thin-section computed tomography. Respir Med. 2013;107(6):904–10.
34. Zhang Z, Zhou L, Min X, Li H, Qi Q, Sun C, Sun K, Yang F, Li X. Long-term follow-up of persistent pulmonary subsolid nodules: natural course of pure, heterogeneous, and real part-solid ground-glass nodules. Thoracic cancer. 2023;14(12):1059–70.
35. Chang B, Hwang JH, Choi YH, Chung MP, Kim H, Kwon OJ, Lee HY, Lee KS, Shim YM, Han J, Um SW. Natural history of pure ground-glass opacity lung nodules detected by low-dose CT scan. Chest. 2013;143(1):172–8.
Acknowledgements
The authors would like to thank all the reviewers who participated in the review and MJEditor (www. www.mjeditor.com) for their linguistic assistance during the preparation of this manuscript.
Funding
This study was supported by the Ministry of Science and Technology of the People’s Republic of China (CN): 2022YFF1203301 and Research Project Plan of Shanghai Municipal Health Commission(20214Y0309).
Author information
Author notes
Wufei Chen and Ernuo Wang contributed equally to this work.
Authors and Affiliations
Department of Radiology, Huadong Hospital, Fudan University Shanghai China, Shanghai, China
Wufei Chen, Pan Gao, Fang Lu, Ernuo Wang, Haiquan Liu & Ming Li
Contributions
Haiquan Liu: Conceptualization; investigation; validation; writing – original draft. Wufei Chen and Pan Gao: Data curation and Formal analysis. Fang Lu and Ernuo Wang: reference review and Investigation.
Corresponding authors
Correspondence to Haiquan Liu or Ming Li.
Ethics declarations
Ethics approval and consent to participate
The Ethics Review Board of Huadong Hospital, Affiliated with Fudan University, approved this retrospective study (No.20230108), and it was performed following the Declaration of Helsinki. The requirement for informed consent was waived.
Consent for publication
Not applicable.
Competing interests
There are no conflicting interests that the authors declare.
Credits: Chen, W., Gao, P., Lu, F. et al. CT texture features of lung adenocarcinoma with HER2 mutation. BMC Cancer 25, 287 (2025). https:// doi.org/10.1186/s12885-025-136 86-z